Usability engineering and risk management for medical devices
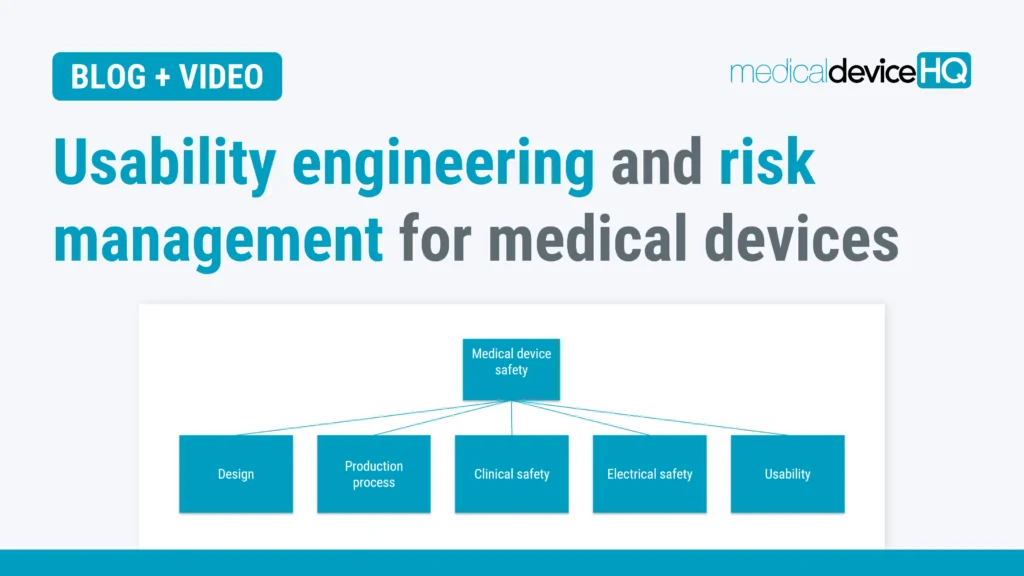
Usability engineering and risk management are connected in many ways, and all steps should be taken to ensure both safe and user-friendly medical devices for the end users.


Usability engineering and risk management are connected in many ways, and all steps should be taken to ensure both safe and user-friendly medical devices for the end users.

It is important to know if a product is a medical device because this is what determines if the MDR applies to that product or not. Therefore, it has a major impact on how the device realisation is done as well as how the product should be approved for the European market.

Summative evaluation is the final evaluation of your product’s user interface and confirmation that it does not result in an unacceptable risk to people. Essentially, it is the stage where you get to see if your whole usability engineering process was successful.

Software system, item and units often cause both discussion and confusion. The blog post will try and clarify these within the terminology.

Risk is something you need to understand thoroughly to deal with risk management properly.
You need good risk management because it is a regulatory requirement, you can avoid killing your patients, and it helps you understand on which areas to focus the most.

If your goal is to avoid being late and spending more money on a project than you have initially planned, keep reading. Project risk management is often a neglected area in project management when in reality, it is among

No matter what kind of medical device you are working on, there are some general requirements related to safety which will apply to your medical device, or the development process of that device. This article will introduce the general

This article will help you in identifying and managing critical components. The term critical components is used to identify safety-related components in the test report form as required by clause 4.8 of the standard. Critical components are an important

In this article, we will take a look at how you can manage safety in a simple and effective way when developing electrical medical devices, while at the same time following the IEC 60601 standards. You will also learn

The instructions for use is not only something which is referred to in many ways, such as user guide, operating manual or labelling. It is also a document which can be instrumental in achieving safety for a medical device.

Take a look at our online Risk Management course on ISO 14971:2019 and online Design Control for Medical Devices course.
These courses are taken by both competent authorities, notified bodies and medical device manufacturers and distributors.
Special launch offer: 349 299 EUR for the online plan & 449 349 EUR for the online lifetime plan.