The course covers the aspects of the IEC 62304 standard, as well as the relationship between IEC 62304 and other standards such as ISO 14971, IEC 60601-1 and IEC 82304-1.
You learn the theory first during the online course, and then apply that theory during the two live virtual classroom sessions.
€ 1099/ seat
| Course length | approx. 16-32 hours (25 CPD points) |
| Next classroom sessions | 1-2 Jul, 2026 |
QUICKSTART
FREE
Average completion time 1-3 hours
Total video duration 32 minutes
Number of quizzes 5
30 days access to the course
Digital course companion
ONLINE
€ 449
Average completion time 9-25 hours
Total video duration 6.5 hours
CPD points 18
Number of quizzes 33
Final exam
Course certificate
Ask the instructor
6 months access to the course and instructor
Digital course companion
ONLINE LIFETIME
€ 549
Average completion time 9-25 hours
Total video duration 6.5 hours
CPD points 18
Number of quizzes 33
Final exam
Course certificate
Ask the instructor
Lifetime access to the course and instructor
Digital course companion
BLENDED
YOU ARE HERE
€ 1099
Average completion time 16-32 hours
Total video duration 6.5 hours
CPD points 25
Number of quizzes 33
Final exam
Course certificate
Ask the instructor
Lifetime access to the course and instructor
Digital course companion
Classroom sessions (2 * 3.5 hours)
Templates
IN-HOUSE TRAINING
CONTACT US
Tailored training to meet your company's needs
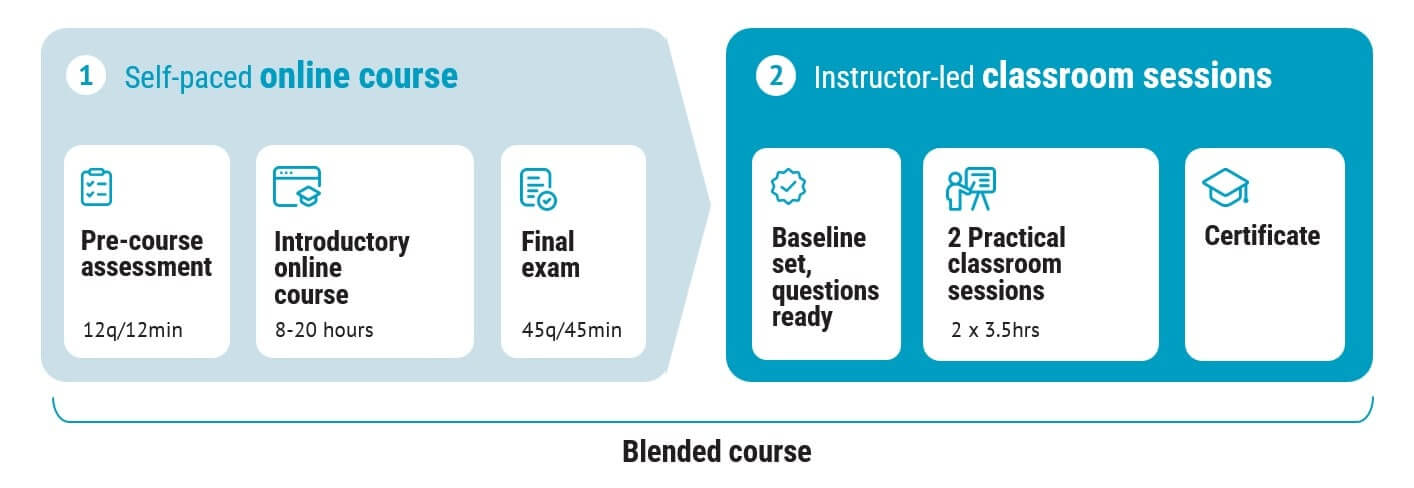
This course consists of two parts:
1. Software for Medical Devices and IEC 62304 online course
2. Two half-day live virtual classroom sessions held by the course creator
First, you complete the popular software for medical devices online course. It covers topics such as how to efficiently plan and document software, getting requirements right, how to approach software architecture and software risk management, how to perform software safety classification correctly and working with SOUP/OTS software. Note: The course does not cover coding.
You can start the online course immediately and you must successfully complete the final exam prior to the classroom sessions. If you have already completed the online course, you can register for the classroom sessions only.
The unique, highly interactive live virtual classroom sessions are facilitated by Christian Kaestner, an expert in this field. During the sessions, you get to apply the theory you have learnt via case studies, workshops, and discussions. There is plenty of time for Q&As, and with a maximum of 8 people per classroom session, all participants have the opportunity to learn first-hand from an industry expert.
A digital course companion is included, as well as time-saving templates on how to implement software for medical devices effectively and efficiently.
We recommend this course to those who are working with software development, such as R&D engineers, quality assurance department and auditors of software development.
Our blended learning courses combine the best of both worlds: online training and two half-day live virtual classroom sessions afterwards.
The online training allows for the flexibility in the way you learn, so that you can learn in a way which suits you best. You can access it whenever and wherever you want, and you can stop, start, and repeat the lessons and quizzes as often as you would like. To help maximise your learning, you can check your knowledge and understanding every step of the way by taking the assessment test beforehand, as well as the checkpoint quizzes that are found throughout the course. Ultimately, you will take a final exam at the end of the course.
The classroom sessions are highly interactive and focuses on applying the theory you have learnt during the online course with the guidance of the expert instructor. There are real life case scenarios to work through and plenty of time for discussions and questions.

Software development for medical devices can be made incredibly abstract if there is too much focus on the intangible requirements instead of how to actually do the work.
This course is very practical; you will be working on realistic tasks relating to the software lifecycle according to IEC 62304.
Gain exclusive up to the minute information by learning from authors of the standards.
Our courses are developed by members of the standardization committees involved at both national and international levels, ensuring you receive first-hand knowledge and insights.
Our instructors are passionate about what they do, bringing their expertise and love for the subject into the learning experience.
Participate in active learning by asking questions directly to the instructor, taking part in group workshops and discussions. This allows you to seek clarification, share insights, apply and transfer knowledge.
The online course comprises a mixture of pre-recorded videos, quizzes, and a final exam. It is self-paced and flexible in that you can start, stop, and repeat as much as you like. You will have lifetime access to the online course (conditions apply).
The Software for Medical Devices and IEC 62304 online course consists of 11 lessons which are outlined below.
You will attend these two half-day live virtual instructor-led classroom sessions via Zoom. The focus is on practicing the new skills, knowledge, and understanding learnt during the online course, in a relaxed environment, with the guidance of an expert instructor. There are real-life case scenarios to work through, and plenty of time for discussions and questions.
The live virtual classes will cover all aspects of the IEC 62304 standard and the relationship between the IEC 62304 and other standards such as the ISO 14971 standard.
The maximum number of people in a classroom session is 8. Please note, the classroom sessions will have participants from both this course and SaMD, IEC 62304 and IEC 82304-1 – Practitioner Training course.
Included is a digital course companion and the following time-saving templates:
The blended course consists of two parts: the online course, and two half-day live virtual classroom sessions.
The online course is made up of carefully scripted pre-recorded videos in order to maximise your learning. The live virtual classroom sessions are virtual face-to-face sessions where you will be interacting with the instructor and the other course participants via Zoom. You are required to be visible on video when taking the classroom sessions.
Yes, you can.
The time spent on the course varies greatly. If you are a beginner or have very high ambitions, you will typically spend more time on it. If you are already experienced in the field, you will probably finish it more quickly. The online course takes 9-25 hours to finish on average.
You do not have to watch the whole course at once. In fact, you can pause, resume, and replay as often as you like.
The two half-day live virtual classroom sessions take approximately 7 hours in total.
You will be receiving a course certificate after completing the final exam. The course certificate is available when you log in to your account. Go to “My Courses”, and scroll down. You will find it under “Your Course Certificates”.
After successfully completing the classroom session, you will receive a course certificate covering both the online course and the classroom session by email.
If you wish to buy 3 or more seats, we invite you to reach out using the contact form to discuss discount options. Please note, if there are more than 5 people from your organisation wising to take the course, consider arranging an in-house course. The price will be lower, and the course can be customised to your needs.
If you complete the final exam within 6 months of enrolment, you will have lifetime access to the online course (conditions apply). If you do not, your course access will expire after 6 months.
Yes, you can download a course companion within the online course. It contains a selection of key slides from the course and is in PDF format.
The maximum number of participants for the live virtual classroom session is 8.
There is no pass/fail score on the exam as such, but you are required to achieve 85% or more on the final exam to be allowed to attend the classroom session. The check-point quiz questions that are available during the online course will be a good model of what you will get on the final exam.
If you do not achieve 85%, the final exam can be reset for an administrative fee of 50 EUR, and you can take it again.
If you have already completed the online course and taken the final exam more than 3 weeks prior to the classroom sessions, and regardless of whether you have achieved >85%, we will reset the final exam for you for free.
You will need to retake the final exam by 3pm CET the day before the classroom sessions. This is because we insist all participants have the information fresh in their mind in order to maximise the learning experience in the live virtual classroom sessions.
The instructor is an expert member of the project team authoring IEC 62304 and also actively participated in the creation of IEC 82304-1.
He has extensive experience of medical device development and a strong dedication to software development. In the software domain he has worked in many roles such as software developer, project manager, auditing and quality management. This ensures the instructor has access to leading-edge knowledge and information, which is the highest qualification available for teaching the course.
We offer a number of payment methods including credit card, GooglePay, ApplePay, PayPal, wire transfer/invoice depending on the country you are buying from. If you cannot find your country on the list at checkout or you are not given wire transfer as a payment method, you can submit your order via our invoice order form.
Medical Device HQ is proud to be an accredited CPD provider, demonstrating our commitment to your continued professional development with high-quality training courses.
A CPD record showcases your professional growth, boosts confidence in your abilities, and can be presented to management for better career opportunities.
As a participant of a CPD certified course, you will receive a certificate awarding you CPD points following completion of the training.
Upon completion of this blended course, you are awarded 25 CPD points.
Your course certificate will display the exact number of CPD points. Read more.




IMPORTANT – The course will be associated with the account that the purchase is made from. Are you taking the course or is someone else?
Choose your course options below
IMPORTANT – The course will be associated with the account that the purchase is made from. Are you taking the course or is someone else?